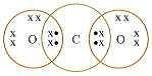
1: What would be the electron dot structure of Carbon dioxide which has the formula CO2?
Ans: Electron dot structure of CO2 is
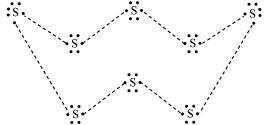
2: What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur? (hint − the eight atoms of sulphur are joined together in the form of a ring.)
Ans: Electron dot structure of a sulphur molecule
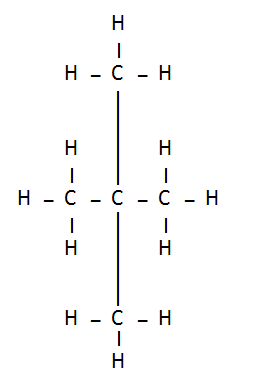
3: How many structural isomers can you draw for pentane?
Ans: Three structural isomers are possible for pentane.
Pentane → C5H12
(a) N- Pentane
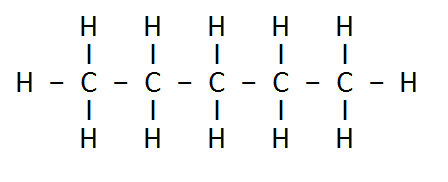
(b) Iso – Pentane
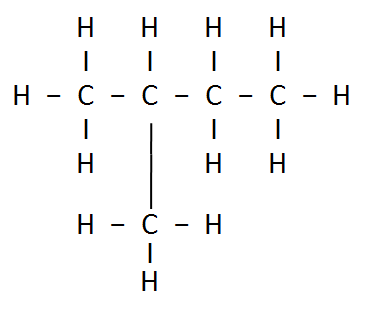
(c) Neo Pentane
4: What are the two properties of Carbon which lead to the huge number of Carbon compounds we see around us?
Ans: The two features of Carbon that give rise to a large number of compounds are as follows:
(i) Catenation: It is the ability to form bonds with other atoms of Carbon.
(ii) Tetravalency: with the valency of four, Carbon is capable of bonding with four other atoms
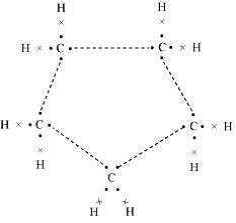
5: What will be the formula and electron dot structure of Cyclopentane?
Ans: The formula for Cyclopentane is C5H10. Its electron dot structure is given below.
6: Draw the structures for the following compounds.
(i) Ethanoic acid (ii) Bromopentane*
(iii) Butanone (iv) Hexanal Are structural isomers possible for Bromopentane?
Ans: (i) Ethanoic Acid (CH3COOH)
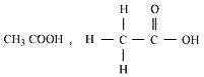
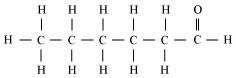
(ii) Hexanal (C5H11CHO)
(iii) Butanone (C2H5COCH3)
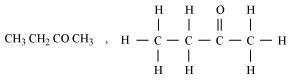
(iv) Bromopentane (C5H11Br)
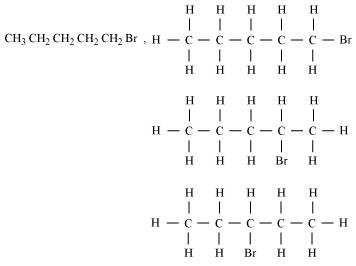
Yes Structural isomers are possible for Bromopentane
7:How would you name the following compounds?
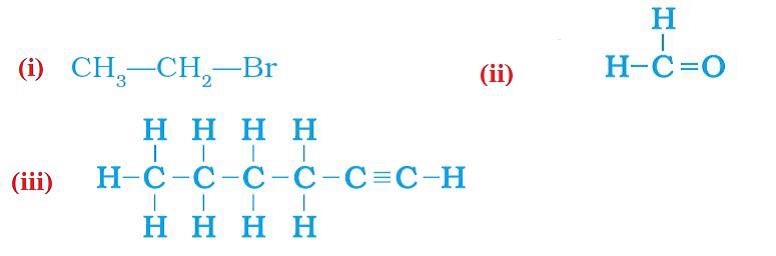
Ans:
(i) Bromoethane (ii) Methanal (iii) Hexyne
8: Why is the conversion of ethanol to Ethanoic acid an oxidation reaction?
Ans: Since the conversion of ethanol to Ethanoic acid involves the addition of oxygen to ethanol. So it is an oxidation reaction.
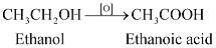
9: A mixture of oxygen and Ethyne is burnt for welding. Can you tell why a mixture of Ethyne and air is not used?
Ans: When Ethyne is burnt in air, it gives a sooty flame. This is due to incomplete combustion caused by limited supply of air. However, if Ethyne is burnt with oxygen, it gives a clean flame with temperature 3000°c because of complete combustion. This Oxy-acetylene flame is used for welding. It is not possible to attain such a high temperature without mixing oxygen. This is the reason why a mixture of Ethyne and air is not used.
10:
(i)
(a) Litmus Test:
Take 2ml of alcohol and carboxylic in a test tube. Put 1 drop of blue litmus in each test tube. The liquid that shows no effect is alcohol. The liquid that turns red is Carboxylic acid.
(b) Sodium bicarbonate Test
Take a small amount of each compound add aqueous NaHCO3. The compound that produces brisk effervescence due to the evolution of CO2 is carboxylic acid & the one that shows no effect is alcohol.
(ii): What are oxidising agents?
Ans: Some substances such as alkaline potassium permanganate etc are capable of adding oxygen to others. These are known as oxidising agents.
11: Would you be able to check if the water is hard by using a detergent?
Ans: Unlike soap, they do not react with Calcium and Magnesium ions present in hard water to form scum. They give a good amount of lather in both hard or soft water. This means that detergents can be used in both soft and hard water. Therefore, it cannot be used to check whether the water is hard or not.
12: People use a variety of methods to wash clothes. Usually, after adding the soap, they beat the clothes on a stone, or beat it with a paddle, scrub with a brush or the mixture is agitated in a washing machine. Why is agitation necessary to get clean clothes?
Ans: A soap molecule has two parts namely hydrophobic and hydrophilic. With the help of these, it attaches to the grease or dirt particle and forms a cluster called micelle. These micelles remain suspended as a colloid. To remove these micelles (entrapping the dirt), it is necessary to agitate clothes.

